UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
______________________
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
______________________
Date of Report (Date of earliest event reported): March 30, 2021
| (Exact name of registrant as specified in its charter) | ||
| (State or other jurisdiction of incorporation) | ||
| (Commission File Number) | (I.R.S. Employer Identification Number) | ||||
| (Address of principal executive offices) | (Zip Code) | ||||
Registrant’s telephone number, including area code: (574 ) 268-6379
| Not Applicable | ||
| (Former name or former address, if changed since last report) | ||
Securities registered pursuant to Section 12(b) of the Act:
| Title of Each Class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) | |||||
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) | |||||
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) | |||||
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) | |||||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 under the Securities Act (17 CFR 230.405) or Rule 12b-2 under the Exchange Act (17 CFR 240.12b-2).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act ☒
Item 7.01. Regulation FD Disclosure.
The executive officers of OrthoPediatrics Corp. have several upcoming presentations to representatives of investors and analysts. The officers intend to use the material filed as Exhibit 99.1 herewith, in whole or in part, as part of those presentations.
The information in this Item 7.01, including the information incorporated by reference herein from Exhibit 99.1, is furnished pursuant to Item 7.01 of Form 8-K and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or incorporated by reference in any filing under the Securities Act of 1933, as amended, or the Exchange Act, except as shall be expressly set forth by specific reference in such a filing.
Item 9.01. Financial Statements and Exhibits.
| (d) | Exhibits | |||||||
| Exhibit No. | Description | |||||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). | |||||||
* * * * * *
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| OrthoPediatrics Corp. | ||||||||
| Date: March 30, 2021 | By: | /s/ Daniel J. Gerritzen | ||||||
| Daniel J. Gerritzen, General Counsel and Secretary | ||||||||
- 2 -

OrthoPediatrics Corp. April 2021 Mark Throdahl, CEO Fred Hite, COO & CFO Dave Bailey, President

Disclaimer Forward‐Looking Statements This presentation includes "forward‐looking statements" within the meaning of U.S. federal securities laws. You can identify forward‐looking statements by the use of words such as "may," "might," "will," "should," "expect," "plan," "anticipate," "could," "believe," "estimate," "project," "target," "predict," "intend," "future," "goals," "potential,” "objective," "would" and other similar expressions. Forward‐looking statements involve risks and uncertainties, many of which are beyond OrthoPediatrics’ control. Important factors could cause actual results to differ materially from those in the forward‐looking statements, including, among others: the risks related to COVID‐19, the impact such pandemic may have on the demand for our products, and our ability to respond to the related challenges; and the risks, uncertainties and factors set forth under "Risk Factors" in OrthoPediatrics’ Annual Report on Form 10‐K filed with the SEC on March 11, 2021, as updated and supplemented by our other SEC reports filed time to time. Forward‐looking statements speak only as of the date they are made. OrthoPediatrics assumes no obligation to update forward‐looking statements to reflect actual results, subsequent events, or circumstances or other changes affecting such statements except to the extent required by applicable securities laws. Use of Non‐GAAP Financial Measures This presentation includes the non‐GAAP financial measure of Adjusted EBITDA, which differs from financial measures calculated in accordance with U.S. generally accepted accounting principles (“GAAP”). Adjusted EBITDA in this release represents net loss, plus interest expense, net plus other expense, provision for income taxes (benefit), depreciation and amortization, stock‐based compensation expense, fair value adjustment of contingent consideration, acquisition related costs and accrued legal settlement costs. Adjusted EBITDA is presented because the Company believes it is a useful indicator of its operating performance. Management uses the metric as a measure of the Company’s operating performance and for planning purposes, including financial projections. The Company believes this measure is useful to investors as supplemental information because it is frequently used by analysts, investors and other interested parties to evaluate companies in its industry. The Company believes Adjusted EBITDA is useful to its management and investors as a measure of comparative operating performance from period to period. Adjusted EBITDA is a non‐GAAP financial measure and should not be considered as an alternative to, or superior to, net income or loss as a measure of financial performance or cash flows from operations as a measure of liquidity, or any other performance measure derived in accordance with GAAP, and it should not be construed to imply that the Company’s future results will be unaffected by unusual or non‐recurring items. In addition, the measure is not intended to be a measure of free cash flow for management’s discretionary use, as it does not reflect certain cash requirements such as debt service requirements, capital expenditures and other cash costs that may recur in the future. Adjusted EBITDA contains certain other limitations, including the failure to reflect our cash expenditures, cash requirements for working capital needs and other potential cash requirements. In evaluating Adjusted EBITDA, you should be aware that in the future the Company may incur expenses that are the same or similar to some of the adjustments in this presentation. The Company’s presentation of Adjusted EBITDA should not be construed to imply that its future results will be unaffected by any such adjustments. Management compensates for these limitations by primarily relying on the Company’s GAAP results in addition to using Adjusted EBITDA on a supplemental basis. The Company’s definition of this measure is not necessarily comparable to other similarly titled captions of other companies due to different methods of calculation. 2

Highlights Diversified medical device company focused exclusively on pediatric orthopedics Protected market opportunity: $1.5 billion U.S., $3.3 billion globally (2020 estimates) High U.S. procedure concentration: <300 hospitals and ~1,400 surgeons Focused call point: pediatric orthopedic surgeons are generalists who use all OP products Sustainable competitive advantage: ‐ Broadest pediatric‐specific orthopedic product offering with 35 surgical systems ‐ Strong relationships with pediatric orthopedic surgeons ‐ Deep commitment to clinical education ‐ Sales personnel are a consultative resource who attend surgery Consistent 20+% growth since inception, ex COVID ‐ FY20 revenue of $71.1 million, down 2% ‐ FY19 revenue of $72.6 million, up 26% Recent Orthex and ApiFix acquisitions give OP proprietary, leading‐edge technology in both external fixation and non‐fusion scoliosis markets and expand the Company’s total addressable market Large Market Proprietary Technology Scalable Business 3

COVID‐19 Update During the fourth quarter, our domestic business grew 26% and international agency business grew 51%, supporting the beginning of an international sales turnaround. Product development proceeds at normal pace Maintained set investment to support new product launches, including Orthex and ApiFix Committed to no lay‐offs or base reduction salary cuts to all direct employees Committed to no reductions in financial support of important surgical societies (unlike other industry sponsors) Utilizing DocMatter website to sponsor webinars on pediatric orthopedic care Environment and Company Response 4 Impact on Financials 4Q 2020 sales of $18.9M, down 0.1% vs prior year 4Q 2020 U.S. sales continued strong acceleration, growing 26% year‐over year 4Q 2020 international sales declined 77.7% vs 4Q 2019 Cost savings realized in 2020 used to establish 2021 budget spending

OP Today Treated >200,000 patients since inception 35 surgical systems; ~7,800 SKUs; strong pipeline 6 additional systems from Orthex and ApiFix acquisitions 116 direct employees; 171 focused sales reps2 Global sales organization focused on pediatric orthopedic surgeons in 44 countries1 84 issued patents; 75 patent applications2 Chief Medical Officer is a fellow surgeon Average FDA approval time: < ½ industry average History of stable reimbursement Improving the lives of children with orthopedic conditions Company SnapshotCause 5 A Company Built on a CAUSE Gideon with CMO Emeritus Peter Armstrong, c. 1995. Gideon’s drawing of his girlfriend, 2016. 1 As of January 31, 2021. 2 As of December 31, 2020. Totals apply to patents issued to and/or applied by OrthoPediatrics or one of its wholly‐owned subsidiaries.

Our Key Idea Address orthopedic industry’s lack of focus on product development, clinical education, and sales presence Implants and instruments avoid complications of re‐purposed adult products Product development in collaboration with leading pediatric orthopedic surgeons Dedicated sales support attending surgeries Clinical education programs that build brand loyalty OP’s Market Impact 6 Screws Through Growth Plate Screws Parallel To Growth Plate Superior Clinical Outcomes Children Are Not Small Adults Re‐Purposed Adult Plate OP’s Solution

38% 62% Large and Focused Market Current products target three of the largest categories in Pediatric Orthopedics Pipeline products underway to expand addressable market Trauma & Deformity $609M OP’S $3.3 Billion Current Addressable Global Market1 $1.5 Billion U.S. Addressable Market1 Procedures (%) High Concentration of Pediatric Trauma & Deformity and Scoliosis Procedures Smart Implants $382M Scoliosis $315M Sports Medicine $187M 268 U.S. Hospitals 3,157 U.S. Hospitals 71 Management’s Dec. 2020 updates to IMS data from 2016

Product Line & Growth Diversification $71.1 million sales in 2020, decreased 2% vs prior year ($2.7) million Dec ’20 revenue reduction impacted 2020 growth by negative 4% $72.6 million sales in 2019, increasing 26% Well diversified product sales and sources of growth All products have comparable gross margins 2020 Revenue by Segment (% Total) 8 2020 Revenue by Product Family Revenue by Geography Sports Medicine/ Other Scoliosis Trauma & Deformity 29% 67% 4% 24% 11% 76% 89% 0% 20% 40% 60% 80% 100% 2019 2020 International USA * Excluding ($2.7) million revenue reduction

A Proven Strategy Since 2011 9 Deploy Instrument Sets Sales Focus on Teaching Institutions and High Volume Hospitals Expand Addressable Procedures Expand Clinical Education Programs Goals Accelerate sales growth Develop and acquire novel technologies

10 New Systems & Product Launches (2017‐2018) FIREFLY® Pedicle Screw Navigation Guides (Complementary to RESPONSE Spine System) Tr au m a & D ef or m it y Sc ol io si s Titanium PediPlates® System (Expands physeal tethering offering) Clavicle Plate System (First pediatric specific system) Wrist Fusion Plate System (First pediatric specific system) Medial Patella Femoral Ligament Reconstruction System (Complementary to ACL Reconstruction System) Pediatric Nailing Platform | FEMUR (Expands into adolescent cases) RESPONSE 4.5/4.75/5.0mm System (Maximizes intraoperative flexibility) FireFly S2/Alar PediFlex Advanced Sp or ts M ed ic in e

11 Internal Developments & 2 Select Acquisitions BandLoc DUO System Tr au m a & D ef or m it y Sc ol io si s Launched Feb’19 PediFoot (First pediatric foot system) Launched Nov’19 RESPONSE™ Neuromuscular System Neuromuscular FDA approval Mar’20 PediFoot Expansion Launching 2021 Next Generation Cannulated Screw Systems Launched Sept’19 QuickPack™ Bone Void Filler Launched Dec’19 Large Fragment Cannulated Screw System Launched Mar’20 Orthex AUS / EMEA Launched 2020 / 2021 Orthex (External fixation hardware / software) Acquired June’19 ApiFix MID‐C System (Non‐fusion technology) Acquired Apr’20

Knee (Sports Medicine) Strong Pipeline Expanding Our Addressable Market Spine Foot & Ankle Hip & Long Bone CMF Proximal Humerus Elbow Hand & Wrist Pelvis Clavicle Rib OP Today OP Tomorrow Now Under Development Demonstrated ability to expand portfolio to full array of pediatric surgeries 12 Growing Rods

Leading Edge Systems in Development 13 Emerging surgical trends not being pursued by major spine companies Intervention in patients as young as 10 Reversible, non‐fusion procedures Developing IP portfolio Working with panel of leading surgeons Proof of concept established in 2018 with substantial development in 2019 2 embodiments: (1) scoliosis (2) intramedullary nailing OP will offer significant improvements to current technology Smart Implants Early Onset Scoliosis

Sales Coverage in the U.S. and 44 Countries United States 88.6% of 2020 Revenue 36 Independent Sales Agencies, most of which are exclusive* International 11.4% of 2020 Revenue 14 Independent Sales Agencies + 41 Stocking Distributors* OP’s U.S. business shifted from hybrid model to 100% indirect in 2016 Sales agencies are independent legal entities that take title to and resell product very rarely Converting to agency model in select markets has significantly increased volumes, ASPs, and gross margin Direct in UK, IRE, AUS, NZ, CAN, BE, NL, IT, D, A, CH 14 * Data as of January 31, 2021

Vilex Acquisition 15 Transaction Details Acquisition: In June 2019 OP purchased Vilex1 and its Orthex Hexapod2 system with proprietary point‐and‐click planning software, for $60 million ($50 million cash + $10 million shares) Divestiture: In December 2019 OP sold the adult assets and Orthex license for non‐pediatrics market to Squadron Capital for $25 million cash. Net: Orthex Hexapod investment of $35 million Benefits Expands OP’s Trauma & Deformity business into new segment valued at $200 million globally Expands Trauma & Deformity’s breadth from 60% to 80% of addressable market Increases surgeon reach to limb reconstruction specialists who treat pediatric patients beyond children’s hospitals, generating pull‐through of other products Divestiture allows OP to remain committed solely to pediatrics with cross license rights 1 Vilex generated $6.7 million of revenue in 2018 (most of which was adult) 2 Hexapod had 50% annual revenue growth since FDA clearance in mid‐2016; generated $5.1 million of revenue in 2018

Orthex Advantages 16 Disruptive Technology Construct allows 90° angulation Unique calibrated structs and HA‐coated pins Patented point and click software Significantly simplifies surgery planning and subsequent alignments Dror Paley, MD – Pediatric orthopedic KOL Introduced Ilizarov method in U.S. Defend competitive position and risk Defend other potential acquirers from entering the pediatric space

ApiFix Acquisition 17 Transaction Details Acquisition: In April 2020, OP purchased ApiFix1 and its MID‐C minimally invasive deformity correction system, for 934,768 shares of common stock and $2 million in cash paid at closing, plus milestone payments and an earnout over a period of four years Benefits Expands OP’s Scoliosis business into non‐fusion market, the holy grail of pediatric scoliosis surgery One of only two non‐fusion technologies approved by the U.S. FDA and granted pediatric HDE Least invasive, removable system that acts as an internal brace with motion‐preserving capabilities to avoids permanently limiting range of motion Measurable reductions in surgery time, blood loss, hospitalization, recovery time, and complication rates Extremely high sales/dollar of set inventory Strong IP protection: 46 granted patents and 26 patent applications2 1 ApiFix generated $0.5 million of revenue in 2019 2 As of December 31, 2020 ApiFix Procedure Spinal Fusion
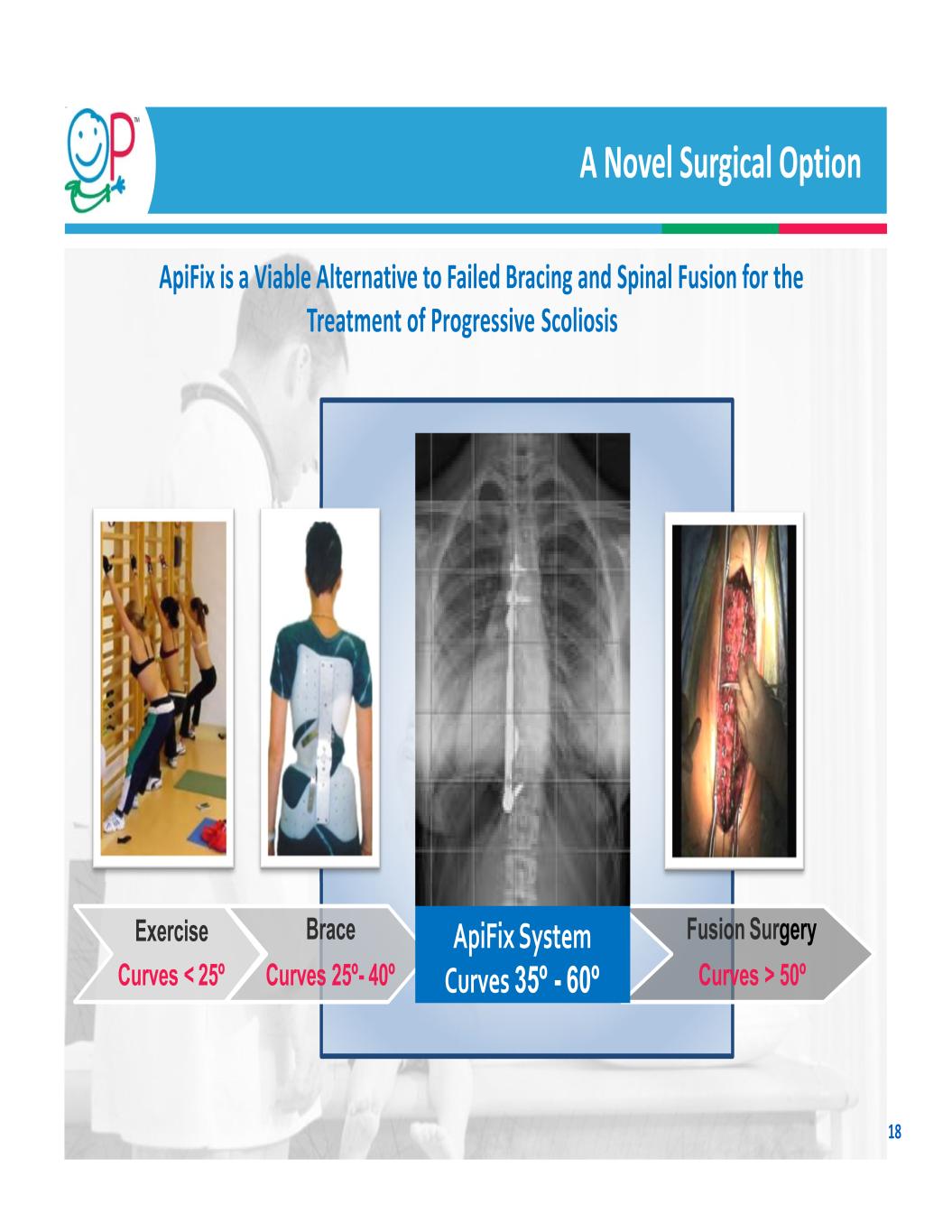
A Novel Surgical Option ApiFix is a Viable Alternative to Failed Bracing and Spinal Fusion for the Treatment of Progressive Scoliosis Fusion Surgery Curves > 50º Exercise Curves < 25º Brace Curves 25º- 40º ApiFix System Curves 35º - 60º 18
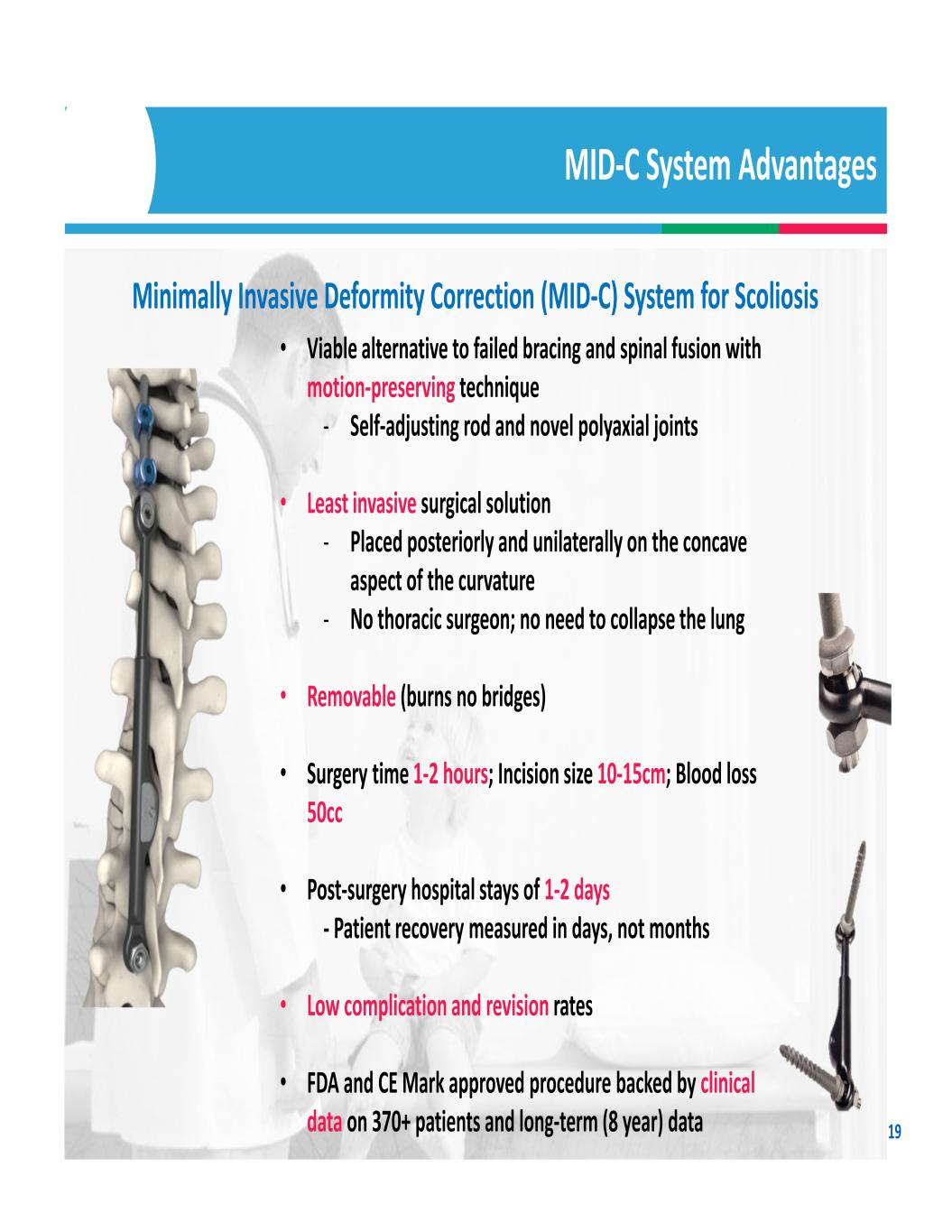
• Viable alternative to failed bracing and spinal fusion with motion‐preserving technique ‐ Self‐adjusting rod and novel polyaxial joints • Least invasive surgical solution ‐ Placed posteriorly and unilaterally on the concave aspect of the curvature ‐ No thoracic surgeon; no need to collapse the lung • Removable (burns no bridges) • Surgery time 1‐2 hours; Incision size 10‐15cm; Blood loss 50cc • Post‐surgery hospital stays of 1‐2 days ‐ Patient recovery measured in days, not months • Low complication and revision rates • FDA and CE Mark approved procedure backed by clinical data on 370+ patients and long‐term (8 year) data 19 Minimally Invasive Deformity Correction (MID‐C) System for Scoliosis MID‐C System Advantages

Product breadth Surgeon relationships Sales and distribution network Clinical education programs Pediatric brand equity Reputation with pediatric orthopedic societies Dynamic culture New Competitors Would Face Formidable Obstacles “The ship has sailed.” Competitive Landscape 20

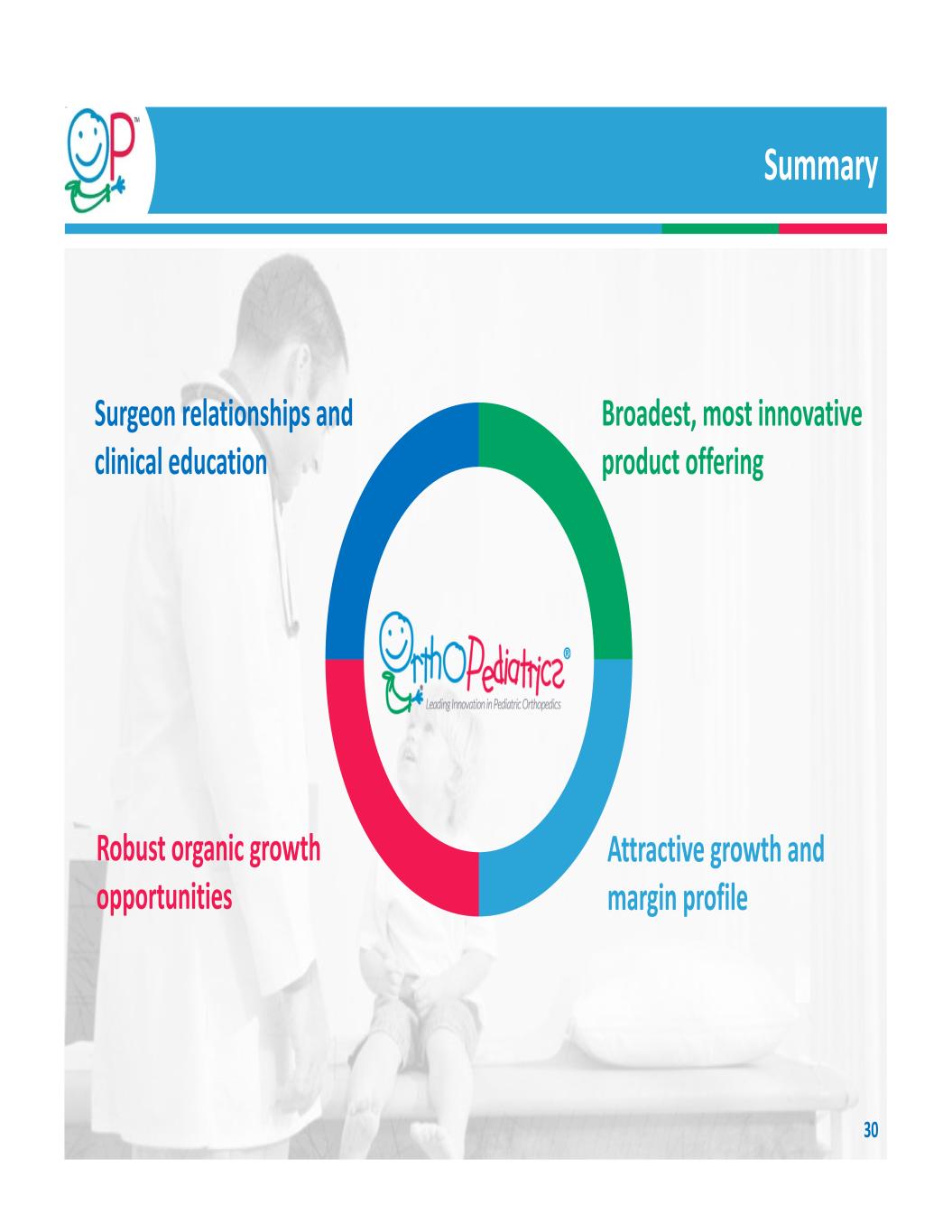
What Does Category Leadership Mean? Surgeon relationships and clinical education Relationships with surgeons who use entire portfolio Major provider of clinical education Leading supporter of surgical societies Custom instruments Attractive growth and margin profile Consistent growth since inception 77% gross margin in FY 2020 History of efficient capital utilization Robust organic growth opportunities $3.3 billion addressable global market Limited focused competition Focused, experienced distribution Instrument set placements drive growth Broadest, most innovative product offering 14 years’ clinical understanding New product pipeline Pediatric Market Gateway for distributed products and joint product developments 21

Financial Review
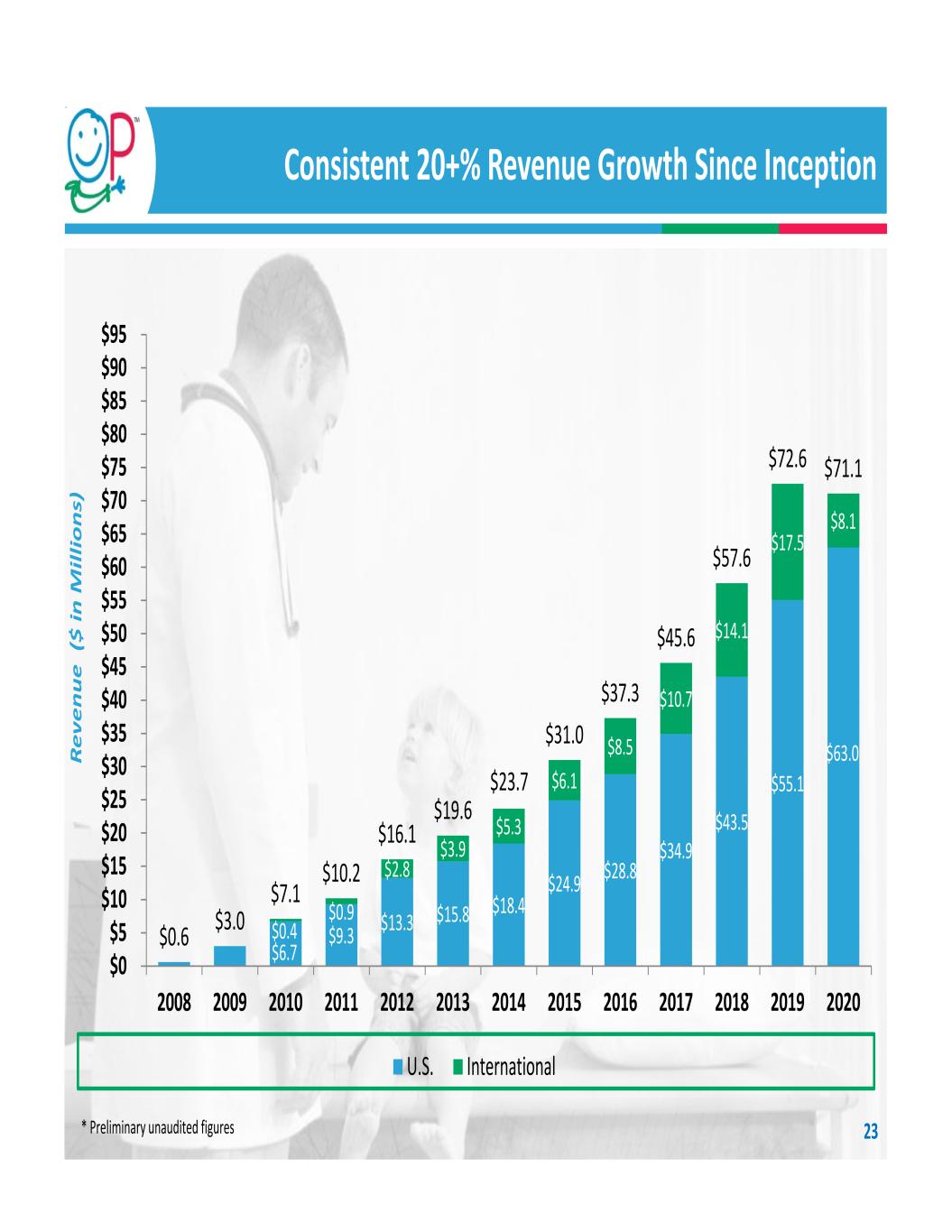
$6.7 $9.3 $13.3 $15.8 $18.4 $24.9 $28.8 $34.9 $43.5 $55.1 $0.4 $0.9 $2.8 $3.9 $5.3 $6.1 $8.5 $10.7 $14.1 $17.5 $8.1 $0.6 $3.0 $7.1 $10.2 $16.1 $19.6 $31.0 $37.3 $45.6 $57.6 $72.6 $71.1 $0 $5 $10 $15 $20 $25 $30 $35 $40 $45 $50 $55 $60 $65 $70 $75 $80 $85 $90 $95 2008 2009 2010 2011 2012 2013 2014 2015 2016 2017 2018 2019 2020 Re ve nu e ($ in M ill io ns ) U.S. International Consistent 20+% Revenue Growth Since Inception 23 $23.7 * Preliminary unaudited figures $63.0
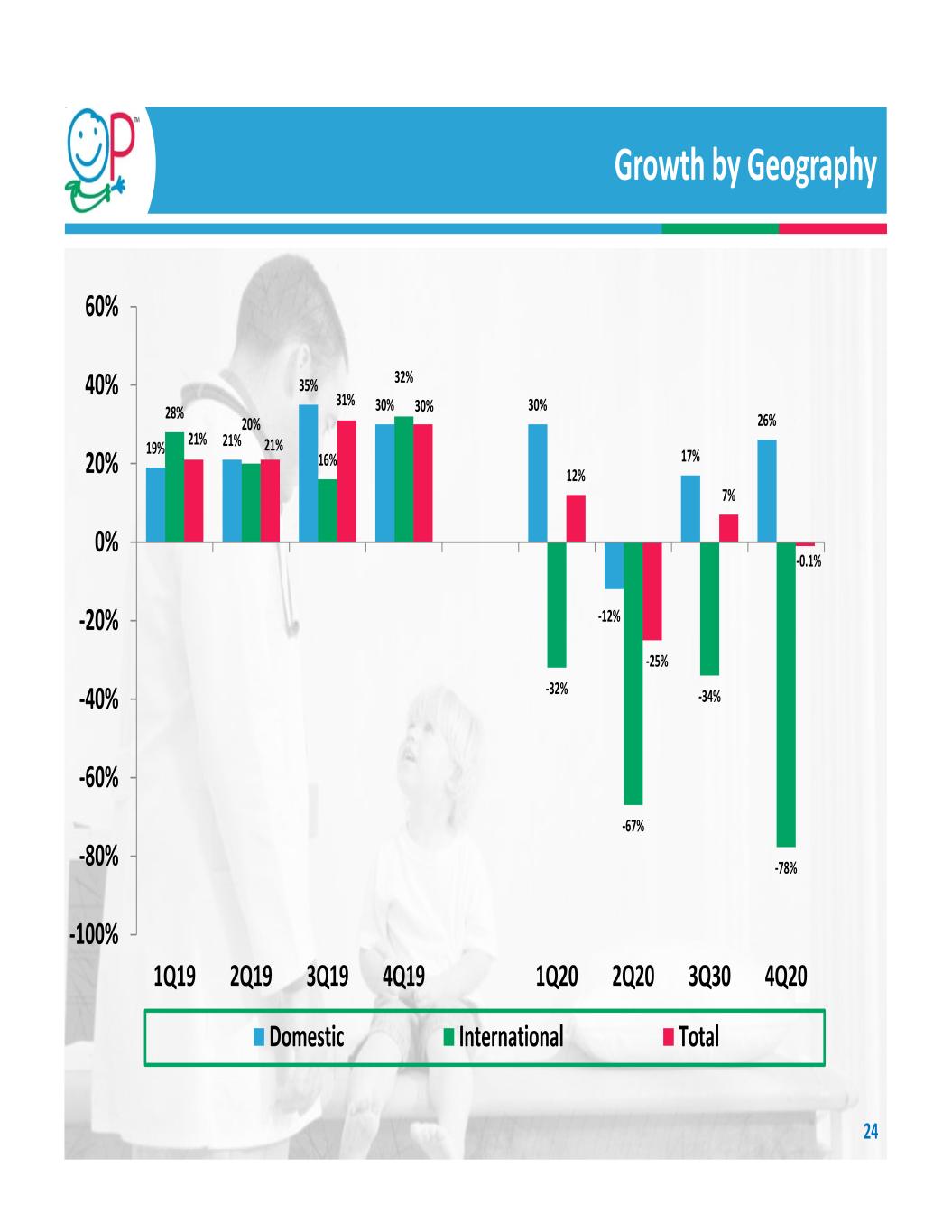
19% 21% 35% 30% 30% ‐12% 17% 26%28% 20% 16% 32% ‐32% ‐67% ‐34% ‐78% 21% 21% 31% 30% 12% ‐25% 7% ‐0.1% ‐100% ‐80% ‐60% ‐40% ‐20% 0% 20% 40% 60% 1Q19 2Q19 3Q19 4Q19 1Q20 2Q20 3Q30 4Q20 Domestic International Total Growth by Geography 24
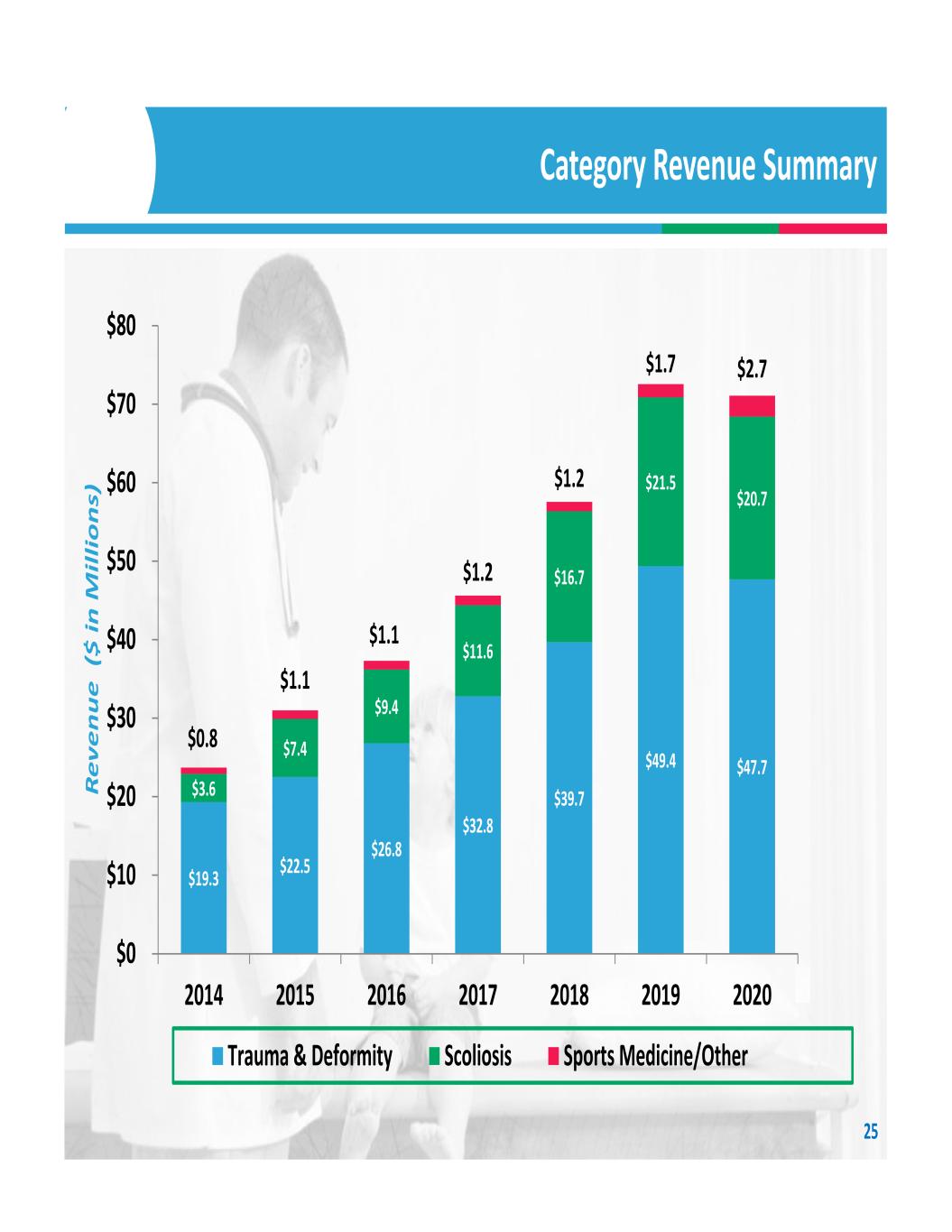
$19.3 $22.5 $26.8 $32.8 $39.7 $49.4 $47.7 $3.6 $7.4 $9.4 $11.6 $16.7 $21.5 $20.7 $0.8 $1.1 $1.1 $1.2 $1.2 $1.7 $2.7 $0 $10 $20 $30 $40 $50 $60 $70 $80 2014 2015 2016 2017 2018 2019 2020 Re ve nu e ($ in M ill io ns ) Trauma & Deformity Scoliosis Sports Medicine/Other Category Revenue Summary 25
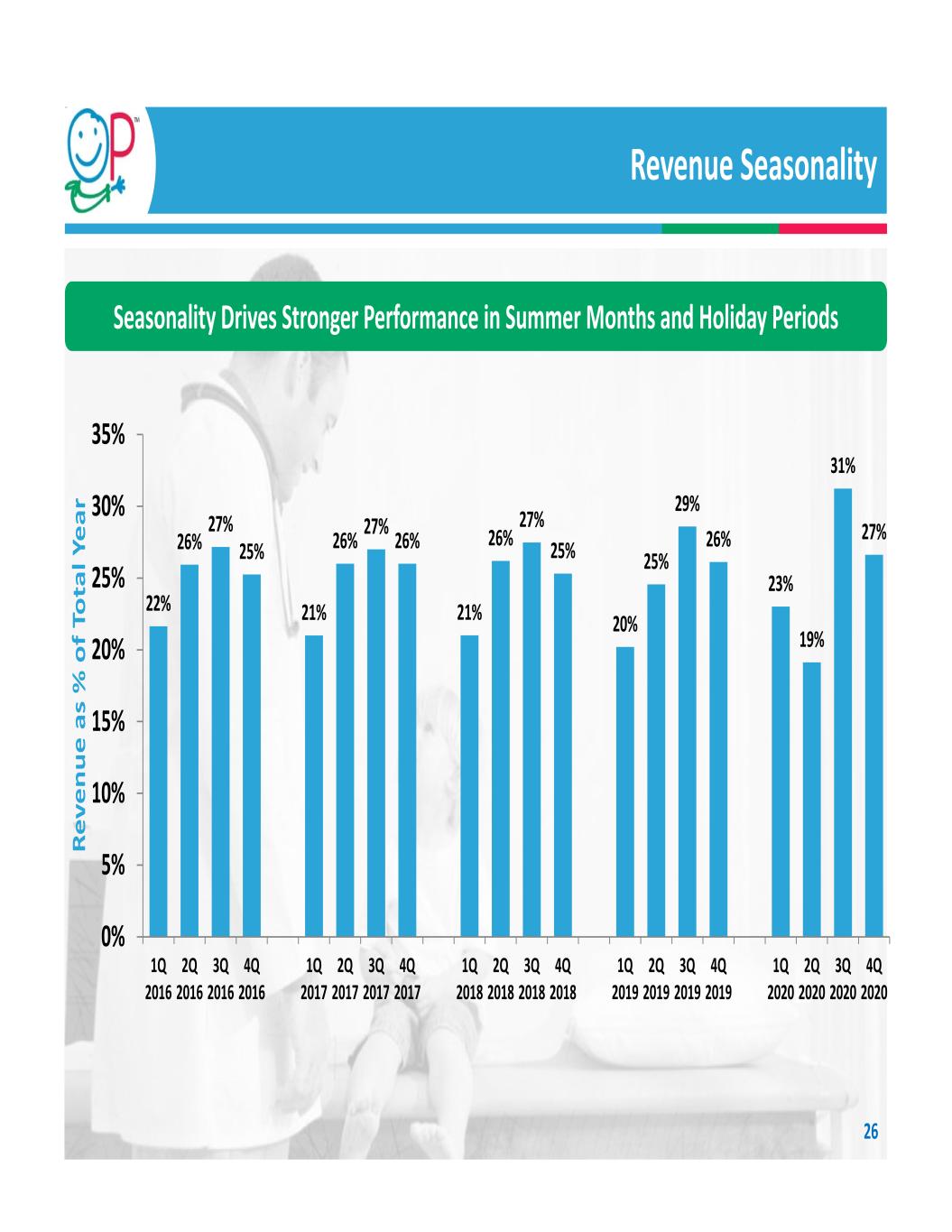
Revenue Seasonality Seasonality Drives Stronger Performance in Summer Months and Holiday Periods 26 22% 26% 27% 25% 21% 26% 27% 26% 21% 26% 27% 25% 20% 25% 29% 26% 23% 19% 31% 27% 0% 5% 10% 15% 20% 25% 30% 35% 1Q 2016 2Q 2016 3Q 2016 4Q 2016 1Q 2017 2Q 2017 3Q 2017 4Q 2017 1Q 2018 2Q 2018 3Q 2018 4Q 2018 1Q 2019 2Q 2019 3Q 2019 4Q 2019 1Q 2020 2Q 2020 3Q 2020 4Q 2020 Re ve nu e as % o f T ot al Y ea r

Income Statement Summary 27 ($ in Millions) 1 Net loss per share attributable to common stockholders – basic and diluted FY 2016 FY 2017 FY 2018 FY 2019 FY 2020 Revenue $37.3 $45.6 $57.6 $72.6 $71.1 Growth % 20% 22% 26% 26% (.01%) Gross Profit $26.4 $34.5 $42.7 $54.6 $55.0 Margin % 71% 76% 74% 75% 77% Operating Expenses $32.5 $40.9 $52.2 $63.7 $81.8 Operating Loss ($6.1) ($6.5) ($9.6) ($9.1) ($26.8) Net Loss ($6.6) ($8.9) ($12.0) ($13.7) ($32.9) Net Loss per Share1 ($7.14) ($5.86) ($0.96) ($0.94) ($1.82)
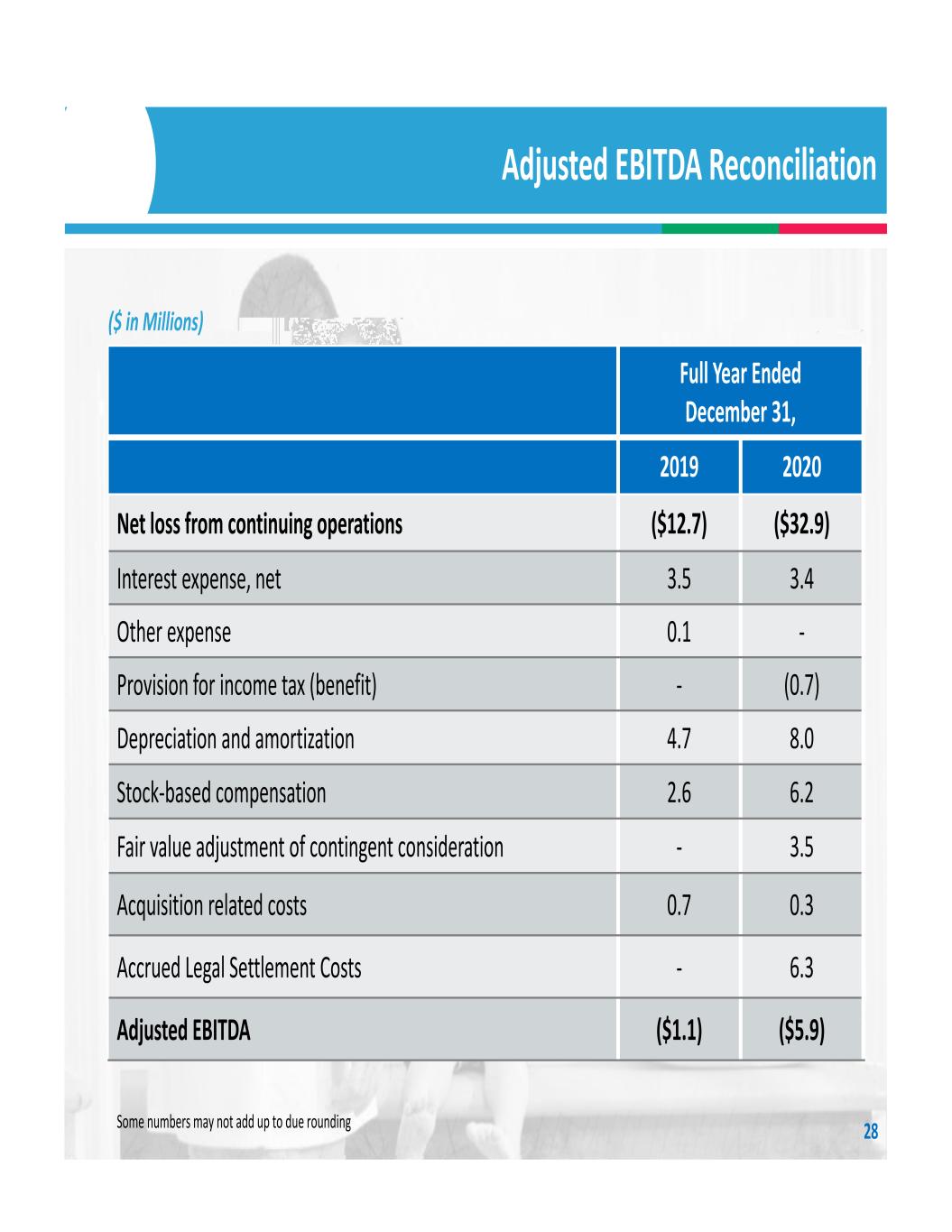
Adjusted EBITDA Reconciliation 28 Full Year Ended December 31, 2019 2020 Net loss from continuing operations ($12.7) ($32.9) Interest expense, net 3.5 3.4 Other expense 0.1 ‐ Provision for income tax (benefit) ‐ (0.7) Depreciation and amortization 4.7 8.0 Stock‐based compensation 2.6 6.2 Fair value adjustment of contingent consideration ‐ 3.5 Acquisition related costs 0.7 0.3 Accrued Legal Settlement Costs ‐ 6.3 Adjusted EBITDA ($1.1) ($5.9) ($ in Millions) Some numbers may not add up to due rounding
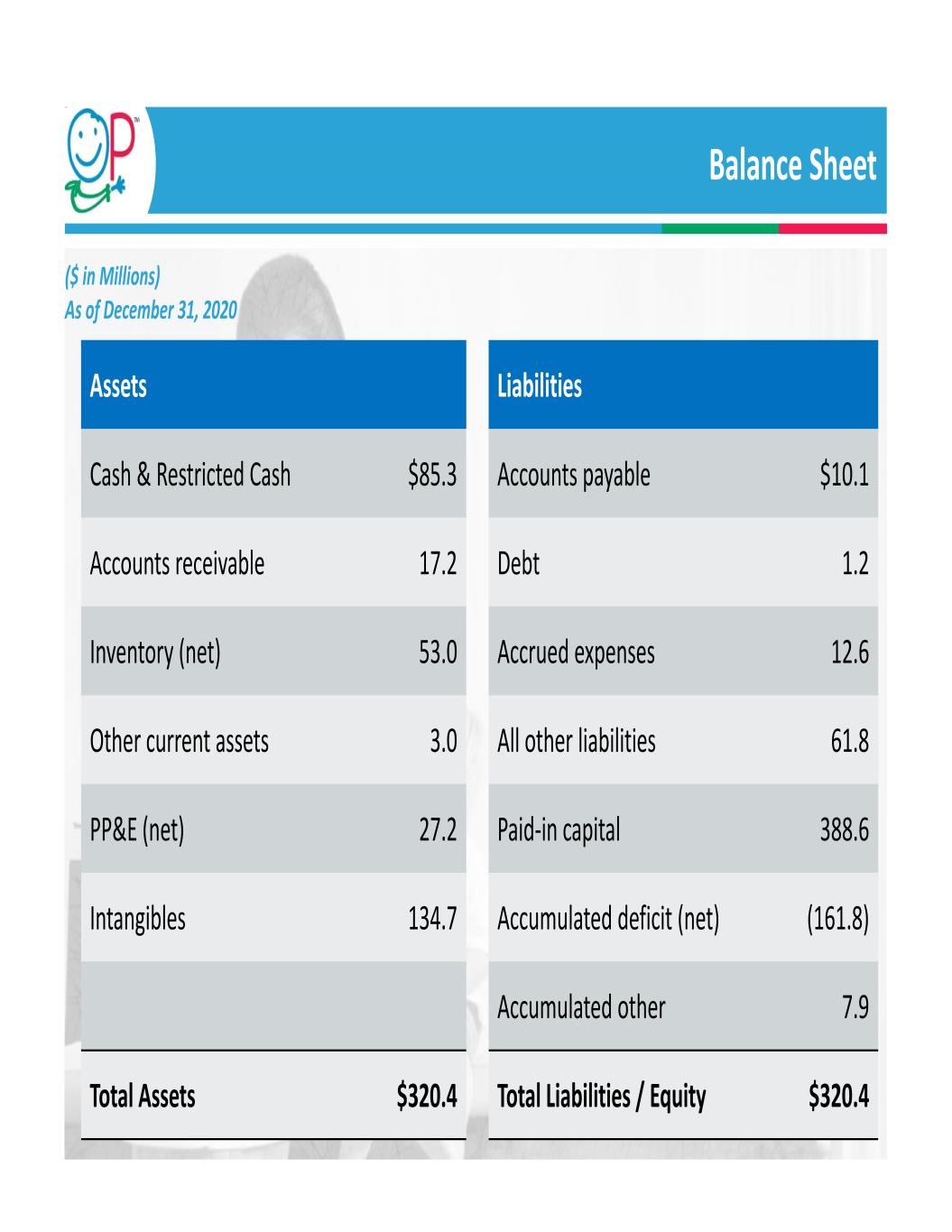
Balance Sheet 29 Assets Cash & Restricted Cash $85.3 Accounts receivable 17.2 Inventory (net) 53.0 Other current assets 3.0 PP&E (net) 27.2 Intangibles 134.7 Total Assets $320.4 ($ in Millions) As of December 31, 2020 Liabilities Accounts payable $10.1 Debt 1.2 Accrued expenses 12.6 All other liabilities 61.8 Paid‐in capital 388.6 Accumulated deficit (net) (161.8) Accumulated other 7.9 Total Liabilities / Equity $320.4
Summary Surgeon relationships and clinical education Attractive growth and margin profile Robust organic growth opportunities Broadest, most innovative product offering 30
